All twelve cultures are under strong selection for rapid growth and all twelve cultures have evolved. One, and only one, of the cultures evolved to utilize citrate as a carbon source (normal E. coli cultures cannot use citrate but it's in the medium as a chelating agent). You can read about the mutations that gave rise to this phenotype in: Lenski's long-term evolution experiment: the evolution of bacteria that can use citrate as a carbon source.
It's important to understand that only one culture evolved this ability. That's because a complex series of mutations had to arise and become fixed in order for the bacteria to be able to use citrate. Because there have been so many generations, every single nucleotide in the genome has been mutated several times in each culture but the probability of just the right mutations happening in just the right order is very low.
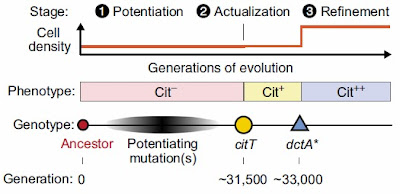
The original analyses were done by Blount et al. (2008) and Blount et al. (2012). They discovered that three different steps were required for efficient citrate utilization. Today, I want to discuss the latest contribution to this study from Quandt et al. (2013).1 It's worth quoting the introduction to their paper since it explains the three stages.
Key innovations in the history of life are often caused by the acquisition of a qualitatively new trait that is "an evolutionary novelty which allows the exploitation of new resources or habitats and thus triggers an adaptive radiation." Such innovations are typically rare and difficult to predict because they result from complex nonadditive (i.e., epistatic) genetic interactions or ecological interactions, within or between species, that develop only over the course of long evolutionary trajectories. Evolution of a new trait can be conceptually divided into three steps: potentiation, actualization, and refinement. First, one or more potentiating events may be necessary to generate a genetic background or environmental conditions that make a new trait accessible to evolution. Genetic potentiation, for example, may involve a period of nonadaptive genetic drift wherein a phenotype stays constant or the accumulation of mutations that are immediately advantageous for reasons unrelated to the new trait. Then, a keystone actualizing mutation or environmental shift may lead to expression of the new trait, possibly by coopting latent changes in a cellular network or physical structure for a new use. Finally, there may be many subsequent opportunities for further refinement mutations that improve an emergent trait so that a newly colonized niche can be fully exploited.The key mutation was a mutation that altered expression of the citT gene allowing uptake of citrate from the medium. This gave rise to a weak Cit+ phenotype that persisted in the population for many generations but did not become fixed. This original mutation occurred in a background of pre-existing mutations that promoted rapid growth.
The weak Cit+ phenotype arose in a single culture after about 31,500 generations. The mutant gene was introduced into earlier ancestors of this strain and into the other eleven strains but it had no effect. This tells you that there was something special about the culture at 35,000 generations—something that "potentiated" the development of the Cit+ phenotype. The citT mutation is the "actualization" step.
The main result of the Quandt et al paper is characterization of a second mutation that enhanced the weak Cit+ phenotype to a strong Cit++ phenotype that took over the entire population. This is the "refinement" step. There are actually two different mutations that lead to enhanced utilization of citrate. The first was duplication of the citT gene leading to production of even more enzymes. This event was characterized by Blount et al. in their 2012 paper.
The second was a mutation in the promoter of the dctA gene leading to an increase in the synthesis of a C4-dicarboxylate transporter. C4-dicarboxylates are important metabolites related to citrate. (Succinate is an example. Recall that succinate and citrate are both part of the citric acid cycle.) Quandt et al. show that this mutation (in dctA) arose after the original citT gene mutation and that the combination of the two mutations is responsible for converting the weak Cit+ to the strong Cit++ phenotype. Carl Zimmer on his blog The Loom describes the result in more detail: Evolution Hidden in Plain Sight.
Although this is an important finding, the most interesting part of the Quandt et al. paper—as far as I'm concerned—is the negative results in the search for potentiating mutations. The authors designed an experiment to analyze large sections of the E. coil genome from the ancestors of the Cit+ strain. Recall that these ancestors carried mutations than potentiated the development of the phenotype while other cultures did not have these mutations so they couldn't give rise to the utilization of citrate.
There were more than 70 mutations that could have been responsible for potentiation but none of them were uniquely identified with potentiation. I like how the authors describe their conclusions since they are clearly aware of modern evolutionary theory and the possibility that mutations can be neutral or even deleterious. This is a characteristic of the people trained in Lenski's lab and it's a refreshing change.
The question remains: how did the potentiating mutations make the highly advantageous Cit++ phenotype more accessible to evolution in this particular LTEE population? In light of our results that argue against the all-or-none epistasis hypothesis, we propose that the potentiating mutations have a quantitative epistatic effect that makes the two-step mutational path required for evolution of the complex Cit++ phenotype from Cit– cells more likely to be realized in the context of an evolving population. Both the evolved citT and dctA alleles may have deleterious or nearly neutral effects on fitness when they are added alone to the ancestral strain or most genetic backgrounds that existed in the LTEE. In this case, weakly Cit+ variants with the citT mutation would rapidly be lost from the population after they arose, due to competition with other alleles that are beneficial for glucose utilization. In fact, however, genotypes with the citT mutation persisted in the population for >1,500 generations, and a small, but significant, fitness benefit was found when the citT allele was added to a Cit– isolate believed to have the potentiating mutations.The important lesson here is that evolution can be highly contingent and largely accidental. The mutations required to "potentiate" the novel pathway may have been neutral or even deleterious. We need to stop thinking that novel traits can be explained solely by gradualistic natural selection operating sequentially on single alleles.
Therefore, on the basis of our results with dctA*, we hypothesize that potentiation may have altered central metabolism in some way that was directly beneficial for growth on glucose but fortuitously increased the supply of C4-dicarboxylates available to power citrate import by the CitT transporter, such that the activating citT mutation became slightly beneficial, rather than neutral or deleterious. This change could have enabled weakly Cit+ lineages to persist in the population long enough to pick up dctA* or other refinement mutations that yielded the highly beneficial, self-sufficient Cit++ phenotype. Assessment of the fitness effect of the citT mutation at various points along the lineage that achieved Cit++ in the future may provide insight into the identity of the potentiating mutations. Finally, it is also possible that the initially very subtle benefit of the Cit+ phenotype depended to some extent on the ecological interactions involving differential uptake and secretion of nutrients by cells in this genetically diverse population.
Not only is evolution more complex than that but it can also explore a much greater range of possibilities than the simple model would suggest.
1. Many Sandwalk readers will recognize one of the authors. It's "deaddog" from talk.origins.
Blount, Z.D., Borland, C.Z. and Lenski, R.E. (2008) Historical contingency and the evolution of a key innovation in an experimental population of Escherichia coli. Proceedings of the National Academy of Sciences 105:7899-7906. [doi: 10.1073/pnas.0803151105]
Blount, Z.D., Barrick, J.E., Davidson, C.J. and Lenski, R.E. (2012) Genomic analysis of a key innovation in an experimental Escherichia coli population. Nature 489:513-518. [doi: 10.1038/nature11514]
Quandt, E.M., Deatherage, D.E., Ellington, A.D., Georgiou, G., and Barrick, J.E. (2013) Recursive genomewide recombination and sequencing reveals a key refinement step in the evolution of a metabolic innovation in Escherichia coli. Proc. Natl. Acad. Sci. published online December 2013 [doi: 10.1073/pnas.1314561111]

24 comments :
It's disappointing that they couldn't pin down which of the 70 possible mutations was/were the actual potentiating mutation. Probably a set of them...
I think of evolution in terms of a curved fitness landscape. A potentiating mutation is like movig horizontally on a landscape (i.e. neutral mutation) where may move to a region with higher or lower slope or curvature, even if your "altitude" is the same. You can imagine this as moving perpendicularly along a saddle structure. f(x) stays the same, but df/dx changes.
If you're initially in a region where df/dx is zero or negative in both directions, you have no ability to do a beneficial mutation. But neutral evolution may move you horizontally to a region where f(x) is the same, but df/dx is now positive. Thus, you now have the potential for beneficial mutations.
We only think of mutations in terms of changes in f(x), which is our mistake. We also have to think in terms of mutations causing changes in the slope df/dx, or the curvature d^2f/dx^2. That's the direction of the 21st century, at least the first half of the 21st century, in evolutionary theory.
It's like the difference between Euclidean geometry and General Relativity, by adding considerations of curvature in fitness space.
Years ago, I saw a diagram of something which was basically an energy well. The bottom of the well represented the global energy minimum. The walls of the well, however, contained local energy minima. An object falling into this well is drawn toward the bottom, but is constrained by the presence of these local minima which must be overcome, just as activation energy is a barrier to a chemical reaction. It helps me to picture this when thinking about evolution.
The metaphor of energy wells is appropriate for protein folding because we know that there IS an energy minimum for a given polypeptide chain and we know that the folded protein will reach it.
Fitness landscapes are entirely different. For any given population we don't even know IF there are ftness peaks, let alone where they are and how high they are. (Same applies to fitness holes.) Furthermore, each fitness lanscape only applies to specific traits, such as ability to use glucose and/or citrate as a carbon source, and not to other traits, such as cell size, mutation rates, and rate of translation.
A population could be on a peak for one trait and a trough for another and their interactions are far to complicated for a simple landscape model. Furthermore, as the population evolves the height and position of the landscape peaks changes. New peaks form and old ones disappear. It's a dynamic lanscape with very little meaning.
We need to stop thinking about fitness lanscapes. It's a metaphor that has outlived its usefullnes.
The result suggests to me a revision of punctuated equilibrium, to be called punctuated potentiation, or something. What was considered equilibrium is now a longish period with a swirl of nearly neutral mutations and drift. Now and then, this sets up conditions for a mutation to become sufficiently favorable to be selected. Once that happens, the positive selection improves the result in relatively less time (15 fold less in this case), making for a punctuation point.
You make one valid and one invalid criticism.
Furthermore, each fitness lanscape only applies to specific traits, such as ability to use glucose and/or citrate as a carbon source, and not to other traits, such as cell size, mutation rates, and rate of translation.
A population could be on a peak for one trait and a trough for another and their interactions are far to complicated for a simple landscape model.
But here you seem to be assuming the fitness landscape only has one dependent variable, x. If you want multiple traits, the fitness landscape is in a higher dimensional space with multiple dependent variables: x, y. Higher spaces can represent interactions and correlations between variables.
as the population evolves the height and position of the landscape peaks changes. New peaks form and old ones disappear. It's a dynamic lanscape with very little meaning.
This is correct, as the environment or competing populations (e.g. predators or prey) change, the fitness landscape has to undulate, so it's variable in time. So it's f(x,y,t) not f(x,y).
However, the concept of "potentiating mutations" must be made mathematical. Moreover, I would argue that is is directly related to Gould's concept of "spandrels", with the spandrel being the potentiating mutation (OK, not a good analogy if the potentiating mutations is neutral) and the painting on the spandrel being the later adaptation contingent on what went before. Gould wrote his paper decades ago. It's time to make these ideas mathematical.
So how do we describe a "potentiating mutation" mathematically? I would describe it as a change from x1 to x2 where
f(x2,y) >= f(x1,y),
(e.g. neutral or slightly deleterious), but
|df(x2,y)/dy| > |df(x1,y)/dy|
You got a better idea? Take a shot.
Errata: should be f(x2,y) <= f(x1,y)
Great post, I learned a lot. But I believe that your second to last conclusion, about the contingent nature of evolution, is a question of perspective. Yes, within the confines of this experiment there was only one lineage that happened to acquire the ability to use citrate, and compared to the many others who didn't it looks awfully lucky for getting just the right potentially neutral mutations.
But zoom out a bit and ask yourself: given thousands of species of bacteria competing over millions of years, how likely is it that none of them will evolve to use citrate? Put that way, and given enough time and and enough material to work on, evolution looks much less contingent.
It is the same with rewinding the tape for all of evolution. Of course there would not necessarily be something that looks just like birds, much less going through exactly the same evolutionary innovations in the same order, but who can seriously doubt that there would be winged animals given the massive advantage that such an adaptation confers?
Me [raising hand]. Archaebacteria, eubacteria and eukaryotes are widely reputed to represent 3 different types of cells. Let's suppose that each one traces back to an ancestor 3 or 4 billion years ago. Now, play the tape forward-- how many of those 3 lineages can count, among their descendants, creatures that fly by flapping wings?
I like the way Richard Dawkins explains contingency in The Blind Watchmaker. He discusses the possibility of evolving "angels"—mammals with wings on their back but also with arms and hands that can do useful work (pp. 308-312). I think we can all agree that this would give these organisms a massive selective advantage. Where are the angels?
It's possible that if we rewound the tape of life we would see pigs that can fly. It's also possible that there wouldn't be any mammals at all and the only large animals would be amphibians or reptiles. It's possible that the alternative Earth would be dominated by highly intelligent crabs or house flies.
I agree with Stephen Jay Gould. Bacteria are the dominant life form on this planet. They have no need of animals and I suspect that most replays of the tape of life would not produce animals.
We are so familiar with our existing world that we find it difficult to imagine any other possibilities. We are imbued with adapationist explanations for all species so we tend to see them all as inevitable products of evolution. What we don't see are all those species than could have evolved but didn't. We also tend to ignore the role of chance and accident in evolution because the only thing anyone wants to talk about is natural selection and adaptation.
Imagine that the mutations leading to the evolution of placenta had never happened. The world would be full of marsupials and there might be highly intelligent kangaroos posting on the internet. Would they be able to imagine any other way of giving birth and nurturing a growing fetus?
Imagine that the mutations leading to the evolution of placenta had never happened. The world would be full of marsupials and there might be highly intelligent kangaroos posting on the internet. Would they be able to imagine any other way of giving birth and nurturing a growing fetus?
And what if the species to develop intelligence was one using external fertilization? Would they have invented the virgin birth tale? :)
The IDers should be all over this with a research program to answer the question: Did the designer intervene to cause this sequence of genetic events?
~~ Paul
Placentas are perhaps a bad example, since they've been evolved many times, mostly in fish. And marsupials have them too. But the point still stands: some evolutionary paths are commonly found, others not. There are factors other than mutation frequency, though. Genetic and environmental background are also crucial.
Larry,
What convinces me of a certain inevitability is that (1) there are so many cases of parallel evolution, of convergence and (2) if there is an unexploited resource or unused habitat it provides an enormous opportunity to those who can utilize or move into it.
There are two counter-arguments. The first one claims that the groups that have converged on the same solution already had the same prerequisites in place when they diverged. Of course one can always say that but it would effectively mean that any possible observation is interpreted as fitting the contingency thesis, thus making it unscientific. And in some cases one would have to claim that all the contingency was already played out at the stage where animals were limbless tiny worms, which is really stretching the idea.
The second is to point at a few cases where no convergence happened, where something evolved only once. Obvious examples are vertebrates, land plants and eukaryotes. The problem here is that I do not see how any of the relevant characters or adaptations can be considered difficult. Vertebrates have an interior skeleton. Land plants have learned to grow on dry land. Eukaryotes have invented a few extra membrane folds and multiple linear chromosomes, and acquired one or two types of endosymbiontic bacteria (and that latter event happened several times in the history of life so it cannot be rare and difficult).
Because these cases are far outweighed by the many cases of convergence, it seems more plausible to explain the few singular cases with the first successful group distorting the fitness landscape: once the first lineage of algae evolved an epidermis, vascular tissue and roots, all late-comers would find the dry land occupied by competitors that are already too well-adapted for them to even try.
But that does not mean that, say, red algae couldn't become land plants if the green algae had not already taken that seat, nor would it mean that such hypothetical red land plants would have any morphospace available that does not include leaves, shoots and roots. A large plant without roots wouldn't work. There is simply a limited number of solutions that are viable, that is the point of the idea of convergence.
As for intelligent marsupials etc, that is no problem for me. As I said, a matter of perspective. As far as I am concerned, that is so indistinguishable from intelligent apes as to make no difference. An intelligent fly or crab, now that would be different, but sadly their circulatory system and exoskeleton do not permit these lineages to achieve the necessary body sizes, so on any planet the big animal niche will at some point be filled by something that is pretty much a vertebrate, regardless of the number of limbs or eyes.
An intelligent fly or crab, now that would be different...
But why not an intelligent octopus? If it were not for their suicidal reproductive strategy, which doesn't let them live long, learn more, and teach their children clever tricks...
Well, could be, why not? It is not me who argues that intelligence is so unlikely and based on so much contingency that human sentience is a fluke that may never happen again. It is the people who stress contingency who often make that argument.
The point I would make is merely that intelligent life on land could not be octopuses because, again, you need to have a decent internal skeleton for that.
""What convinces me of a certain inevitability is that (1) there are so many cases of parallel evolution, of convergence and (2) if there is an unexploited resource or unused habitat it provides an enormous opportunity to those who can utilize or move into it."""
Yes, but I think that when most people talk about contingency in the evolution of life, they are not talking about very general characteristics like having wings or fins. Those are expected adaptational outcomes for common problems, hence bats and birds.
You'd most probably get taxa similar to land plants, aquatic plants, animals etc, but how close they would be to existant taxa is another matter. I certainly don't think youl'd see chimpanzees and orangutans again.
"""Did the designer intervene to cause this sequence of genetic events?"""
Of course. And IDers already know the answer: the sequence of genetic events was put in motion to produce humans. Chain of Being Revisited and all that.
You can only invoke convergent evolution when you see it. If something has evolved only once, then you simply cannot claim it was certain to evolve due to convergent evolution. Not all traits evolve multiple times.
Human-like intelligence arose only once, as far as we can tell. Therefore it cannot be thought of as an inevitable outcome of evolution. This is also the reason why convergent evolution cannot be used as an argument in support of the view that "God had it all planned". It could only be used so if intelligence arose more than once. But if it had evolved more than once, that would be an even bigger theological disaster when you think about it.
Pedro Pereira,
Clearly there is a wide field here. The extreme position on the one end is that everything had to be very much as it came out, with humans as the end product, leading logically to the expectation of Star Trek type humanoid aliens if there is life on other planets. The extreme position at the other end is that everything is so contingent that we would not even recognize the life on other planets as life. Of course I do not claim the first extreme position, but note that those who say that vertebrates may not have evolved on other planets despite the enormous possibilities that become available to the first wormlet to 'invent' an internal skeleton are perhaps closer to the second extreme than I am to the first.
Georgi Marinov,
I do not claim convergence where there is no convergence, I am an atheist, and I don't think teleology is a thing. The argument runs like this:
1. There are so many cases of blatant convergence (eyes, wings, legs, circulatory systems, legless vertebrates, streamlined bodies in water, maternal care, eusociality, cellular endosymbiosis, leaves, secondary growth, heterospory, ...) that the contingency model is already on shaky ground.
2. The best argument for contingency are the few cases in which a crucial innovation was only acquired once (see the three I listed above, perhaps add muscles).
3. But given that there are so many cases of convergence, perhaps a better explanation for these few cases of unique innovations is that the first lineage to acquire it competitively excludes half-baked latecomers.
I cannot judge the eukaryote case but the one with the vascular plants seems pretty strong. If, say, some brown algae would now 'try' something on the lines of Aglaophyton as a first attempt to colonize dry land, how long do you think they would last against green land plants that already have invented roots and leaves?
That post was not intended to correct you (or anyone specifically), sorry for the misunderstanding. I was just adding a needed (in my opnion) clarification to the discussion
Oh, sorry then.
Alex,
I think we agree in general terms, but the question is what "model" of contingency are we talking about here. As you said, there's a whole spectrum of degrees of contingency, so your statement that "that the contingency model is already on shaky ground" depends entirely on what degree of contingency is being proposed by someone. I don't think there's any "standard contingency model" out there that justifies that comment.
Otherwise, yes, we agree pretty much in general.
I agree with Stephen Jay Gould. Bacteria are the dominant life form on this planet. They have no need of animals and I suspect that most replays of the tape of life would not produce animals.
I very much like Gould's explanation of the evolution of more complex forms of life in Full House. He gives the metaphor of a bush growing next to a house. It will branch off into all possible directions. One direction is cut off by the house. Same with life. It branches off in all directions from its origins - more complex, equally complex - except the one closed off to it, less complex, since that leads quickly to not-life. So life has no tendency toward complexity, it just explores whatever is available; and less complex, insofar as the simplest forms of life are concerned, is not available.
Post a Comment