 We know a lot about protein synthesis and structure. Proteins are made by the translation machinery (ribosomes + factors) as they copy the information in messenger RNA. When they are first synthesized, proteins can be visualized as random coils or even linear molecules consisting of a long string of amino acids joined end-to-end.
We know a lot about protein synthesis and structure. Proteins are made by the translation machinery (ribosomes + factors) as they copy the information in messenger RNA. When they are first synthesized, proteins can be visualized as random coils or even linear molecules consisting of a long string of amino acids joined end-to-end.
Eventually these newly synthesized molecules have to fold into a specific three-dimensional shape that's different for every protein. The diagram on the right illustrates this process for some hypothetical folding pathways.
To a first approximation, the final three-dimensional shape is determined by the amino acid sequence of the protein. The final shape represents the lowest free energy state of the folded protein and this can be represented as a free energy well. Left to their own devices, almost all proteins will eventually reach the bottom of the deepest well that represents the functional state of the protein. (There are exceptions to every rule in biology but this is a very good generality.)
 There are many dips and troughs in the free energy landscape and sometimes proteins get trapped in a local minimum as shown by path B in the diagram on the left. If you wait long enough, the incorrectly folded protein will eventually get out of the local dip and fold into the correct shape. (This depends on an energy of activation.)
There are many dips and troughs in the free energy landscape and sometimes proteins get trapped in a local minimum as shown by path B in the diagram on the left. If you wait long enough, the incorrectly folded protein will eventually get out of the local dip and fold into the correct shape. (This depends on an energy of activation.)
For the majority of proteins, this spontaneous folding is quite rapid. They reach the proper three-dimensional structure in seconds or minutes. For some proteins it may take much longer, especially if the free energy landscape is rugged and has many deep pits. When a spontaneous biochemical reaction is too slow to be useful it usually means that an enzyme is required to speed up the reaction. Recall that the role of enzymes is to accelerate reactions that occur spontaneoulsy—they do not create new reactions.
The "enzymes" that speed up protein folding are called molecular chaperones and they are among the most highly conserved enzymes in all of biology. As you might expect, these ancient enzymes are present in all species. There are several different kinds of chaperones but one of the most common is called HSP70 (heat shock protein of 70kDa). [Heat Shock and Molecular Chaperones] [The Evolution of the HSP70 Gene Family] [Gene HSPA5 Encodes BiP-a Molecular Chaperone].
HSP70 binds to hydrophobic regions of the folding protein preventing it from aggregating with other partially folded proteins and steering it toward the final three-dimensional structure. This greatly speeds up the folding pathway for those proteins that are otherwise slow to fold. Obviously there has been selection for rapidly folding proteins and/or selection for those that can be effectively assisted by chaperones. The genes for other proteins have not survived so what we see today are proteins that can fold rapidly with, or without, the assistance of chaperones.
Ulrich Hartl has just published a nice review of chaperones in Nature (Hartl et al. 2011). It didn't take long for the IDiots to comment. I spotted a posting on Uncommon Descent:Nature Review Article Yields Unpleasant Data For Darwinism, but that's just a link to another blog posting by a British IDiot named Antony Latham: New research on protein folding demonstrates intelligent design. Here's what Antony Latham has to say about chaperones.
The review in the journal Nature does not discuss the origins of these systems but we need to ask a question: how does all this fit with current evolutionary theory? One might think that such complex systems are confined to mammals or at least the higher orders of animals. This would be a mistake however, because chaperones and chaperonins are in bacteria and archaea also. Indeed it would seem that for any cell to function there needs to be not just proteins but, at the same time, these chaperone systems, which are absolutely essential for proper folding and maintenance of proteins. Without such systems, in place already, the cell will not function.All of the common chaperones fold spontaneously without the assistance of any other chaperones. The reason why they are called "heat shock" proteins is because their synthesis is induced when cells encounter high temperature or other conditions that may cause proteins to unfold or become unstable. These rescue chaperones are made in huge quantities under these conditions to help prevent the destruction of normal cellular proteins. If you understand this then you will understand that the chaperones themselves are capable of rapid spontaneous folding. Even if you didn't know the facts this would seem obvious.
Now, as explained, these chaperone systems are themselves made of proteins which also require the assistance of chaperones to correctly fold and to maintain integrity once folded. Chaperones for chaperones in fact. The very simplest of cells that we know of have these systems in place.
Darwinian evolution requires step by step changes in molecular systems, with one step leading to another in a manner that is statistically reasonable to expect from selection of mutant strains. There is no Darwinian explanation however for the evolution of proteins which already have chaperone systems in place to ensure proper function.
This points very strongly to an intelligent origin of these ‘ingenious’ systems found in all of life.
In the beginning, you didn't need chaperones because every protein folded rapidly on its own. Some of these primitive proteins might have been a bit slow to fold so the evolution of the first chaperones was advantageous because it enhanced the rate of folding for these proteins. The chaperones weren't absolutely necessary for survival but they conferred a selective advantage on those cells that had them.
Once chaperones were present, new proteins could evolve that would otherwise have been too slow to fold in the absence of chaperones. Over time, cells accumulated more and more of these slowly folding proteins so that today no cell can survive without chaperones.
What we can't explain is why the IDiots keep putting their foots in their mouths.
Hartl, F.U., Bracher, A., and Hayer-Hartl, M. (2011) Molecular chaperones in protein folding and proteostasis. Nature 475: 324–332. [Nature]
28 comments :
Obviously there has been selection for rapidly folding proteins and/or selection for those that can be effectively assisted by chaperones.
An interesting thing is a co-evolution of chaperones and their substrates. The hardest proteins to fold are among the most conserved: actins, tubulins and, to some extent, myosins. Their chaperones are incredibly complex multisubunits proteins (CCT has 16 different chains). And yet they are finely tuned to their substrates: mammalian versions won't fold in insect cells (and vice versa). At the same time, bacterial homologs of actin (MreB) and tubulin (FtsZ) fold reasonably well and don't require anything as complicated as their eukaryotic counterparts. In the most striking example of the co-evolution and divergence, skeletal myosin can only be folded with any efficiency in differentiated muscle cells. Any other tissue does not have right chaperones in sufficient quantities to fold it.
I'll quibble slightly with your description. The folding speed of chaperones isn't particularly important from a theoretical perspective. Early proteins could still have undergone selection for more rapid folding from very mediocre ancestors. Just the same way the eye could evolve from very mediocre components.
Dr. Moran you posted:
"In the beginning, you didn't need chaperones because every protein folded rapidly on its own."
Is there supporting evidence for your claim about the way things were "in the beginning"?
Or is that just an interesting speculation on your part?
Now, as explained, these chaperone systems are themselves made of proteins which also require the assistance of chaperones to correctly fold and to maintain integrity once folded.
Where did he come up with that?
Dr. Moran you posted:
"In the beginning, you didn't need chaperones because every protein folded rapidly on its own."
Is there supporting evidence for your claim about the way things were "in the beginning"?
Or is that just an interesting speculation on your part?
Here is what Moran posted:
"In the beginning, you didn't need chaperones because every protein folded rapidly on its own. Some of these primitive proteins might have been a bit slow to fold so the evolution of the first chaperones was advantageous because it enhanced the rate of folding for these proteins. The chaperones weren't absolutely necessary for survival but they conferred a selective advantage on those cells that had them.
Once chaperones were present, new proteins could evolve that would otherwise have been too slow to fold in the absence of chaperones. Over time, cells accumulated more and more of these slowly folding proteins so that today no cell can survive without chaperones."
This is a very nice story. If science only required nice stories all would be fine.
But science requires evidence.
Perhaps there is good evidence for this story.
If there is, we would all certainly gain if someone could present that evidence.
Anonymous writes:
Is there supporting evidence for your claim about the way things were "in the beginning"?
Or is that just an interesting speculation on your part?
Oooh, I know: The intelligence of Nature was responsible - even before there was Nature. Oh wait, how can something that doesn't exist have intelligence?
When you speak of the intelligence of Nature, Anonymous, do you mean pre-biological entities also? If not, how do you explain the origin of biology?
Anonymous seems to be predictably enough demonstrating why I quibbled with the description above. There are a number of perfectly reasonable and unsurprising scenarios that could have actually happened, and nothing approaching strong evidence to rule them out. Nonetheless there is support for the idea that reasonably fast folding was an early protein feature and potentially even a near universal feature of stable protein structures to boot.
http://www.pnas.org/cgi/content/abstract/103/45/16623
For an example that points in this direction
Here is a quote from the link that JGB gave us:
http://www.pnas.org/content/103/45/16623.abstract
"Anfinsen showed that the amino acid sequence alone is sufficient to determine a protein's structure, but the molecular mechanism responsible for self-assembly remains an open question, probably the most fundamental open question in biochemistry."
JGB I am glad you gave us that link. It was thinking when I was reading Moran's post that
the molecular mechanism responsible for self-assembly was still an open question (as the article puts it).
That seems pretty significant.
Puts Moran's speculations into perspective.
We should always be on guard whenever we see a post or an article that uses the word "spontaneous".
It means "we don't know how this occurs".
But it does have a nice kind of pseudo-science sound to it.
I'm a bit surprised nobody has pointed out the part about "the higher orders of animals". Here's another fact of evolution: all currently living lineages of organisms are exactly the same age - the branches going back to the last common ancestor of any comparison are exactly the same length.
So it's completely incorrect to assume that any particular group of organisms is more evolved than any other group - those remarkably complex Bacteria growing on your lightswitch have been evolving for exactly the same amount of time as your cat.
****
"Spontaneous" has a pretty solid definition in chemistry, I don't think Dr. Moran has been using it incorrectly.
I did not say Moran used it incorrectly. It means "we don't know".
Or do you think that:
"self-assembly remains an open question, probably the most fundamental open question in biochemistry."
means we know?
We know a great deal about protein folding. Many of my colleagues are working on it and some are developing computer models to mimic the process.
It's a difficult computer problem because even with a small protein of 100 amino acids the calculation of free energy changes requires days of computing time. Nevertheless, using only basic rules of physics and chemistry, and the amino acid sequence, my colleagues can already get good approximations of the final folded state.
There aren't any mysteries. No intelligent designer is required in the algorithm.
Moran appears! After all the questions of mine he has ignored, I began to think he was a mythic creature. Like the unicorn that people here have such a fondness for.
TheBrummel, I logged in to say something similar to what you said, but found you had already said it. I'm posting anyway because I like my formulation of it. :-)
chaperones and chaperonins are in bacteria and archaea also.
The implication is that bacteria are primitive. What he is missing is that they are in fact very highly evolved, nearly four billion year's worth.
There aren't any mysteries. No intelligent designer is required in the algorithm.
I think you mean "Mysteries" like the theologians refer to them. They manage to pronounce the Capital M when they run into a question that they don't want to solve.
There are mysteries, but they are solvable. What "anonymous" wants is an admission that Mystery = Designer, when in reality mystery = "a darn good question and we can find the answer."
"Spontaneous" does not mean "we don't know". It means "this process occurs without the addition of energy from an external source"
"Mystery" means "we don't know".
I like to distinguish between "problem" and "mystery". A problem is something we don't know yet, but is solveable given sufficient time, effort, and innovation. A mystery is something that we don't know, and wish to avoid knowing because knowing would spoil the mystery. Which has always seemed like a ridiculous excuse for not working on something, or for bothering people who do wish to work on it.
TheBrummell said...
"Spontaneous" does not mean "we don't know". It means "this process occurs without the addition of energy from an external source"
Since "you don't know" then you don't know if the process occurs without the addition of energy from an external source.
It is simply an assumption that you are making, that the process occurs without the addition of energy from an external source.
Why are you trying to hide that fact? Are you striving to surpass Moran in the tricks department? You have a long ways to go.
Anonymous writes:
[Quoting TheBrummell] "Spontaneous" does not mean "we don't know". It means "this process occurs without the addition of energy from an external source"
[Anonymous speaking for himself] Since "you don't know" then you don't know if the process occurs without the addition of energy from an external source.
You keep setting new standards for being thick. In every case in which Dr. Moran used the term "spontaneous" in the original post, it is known that the process of folding does not require an external energy source. Thus TheBrummell had it exactly right, and the premise of your response to him ("since 'you don't know'") is, as per usual, flatly incorrect.
Since "you don't know" then you don't know if the process occurs without the addition of energy from an external source.
Did you read anything written by anybody here? Your use of language is confusing, it is not at all clear why you placed three words inside quotation mark, nor is it clear that you can distinguish between somebody (me) saying "this term has this definition" and "this term does not have this definition".
Are you perhaps some fairly sophisticated computer program, anonymous? A program that has some trouble with context and negatives?
I should not have placed the words "you don't know" in quote marks.
My point is that:
Since we don't know*, then we don't know if the process occurs without the addition of energy from an external source.
It is simply an assumption that the process occurs without the addition of energy from an external source.
If you have trouble with this idea please let me know. Or if you want to quibble about terminology you can do that instead.
*the molecular mechanism responsible for self-assembly is still an open question (as the article puts it).
Whats the point Anon? That there's more research to be done in biochemistry?
Nothing here suggests that an alternative "explanation" like ID (notice the correct use of quote marks) would be more fruitful, and nothing suggests that scientific research on this issue will be a fruitless waste of time. Infact, given its track record, the scientific work will almost certainly reveal those mechanisms that we don't know today.
Further, they explain that a huge number of these proteins fold spontaneously. You claim that unless we eliminate all outside sources of energy to fold the protein, that there probably is some outside source.
These proteins fold without assistance from 'chaperon' molecules, and that is confirmed by the free energy calculations. They don't require any outside source to fold them. There is no reason to think that the first organisms had these chaperones, let alone that therefore they were designed.
MAYBE Dr. Moran ignores your questions because they're stupid questions and you don't learn from the answers. Pearls before swine and whatnot.
It is simply an assumption that the process occurs without the addition of energy from an external source.
Ah, this is it! OK.
I disagree. It is not an assumption that the process occurs without the addition of energy from an external source. It is a well-supported-by-direct-evidence hypothesis, that has passed repeated testing.
The exact details of which peptides in a long polypeptide are brought close together first may not be fully described for any protein. That lack of detailed knowledge in no way invalidates the knowledge that some proteins fold spontaneously, that is, reach a stable, energetically-favourable 3-dimensional shape without the addition of energy from an external source such as (for example) another enzyme driven by ATP.
Saying a reaction occurs spontaneously says absolutely nothing about how much is known about the fine details of that reaction. For example, the reaction that turns urea and water into ammonia and carbon dioxide
(NH2)2CO + H2O - 2NH3 + CO2
is catalyzed by the enzyme urease; it also happens without a catalyst but much more slowly.
This reaction proceeds spontaneously: if all of the reactants are present and the conditions of temperature and pressure are inside some boundaries (liquid water below about 70 Celsius), urea is converted to ammonia and carbon dioxide.
To say that this reaction occurs spontaneously says nothing at all about the structure of urease, or about the configuration of atoms in urea, or about what we may or may not know about the structure of the enzyme.
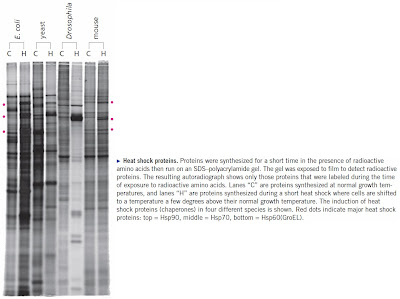
What a beautiful gel! I still remember when you helped me plan it for a paper (we don't really need to mention how many years ago that was).
Here is a quote from the link that JGB gave us:
http://www.pnas.org/content/103/45/16623.abstract
"Anfinsen showed that the amino acid sequence alone is sufficient to determine a protein's structure, but the molecular mechanism responsible for self-assembly remains an open question, probably the most fundamental open question in biochemistry."
I will accept what the published article says:
"the molecular mechanism responsible for self-assembly remains an open question, probably the most fundamental open question in biochemistry."
I am more interested in what the published material says,, than I am in the opinions of people who happen to post on this blog.
Foots in their mouths? What a baffoon! Try finding the proper synchronization of noun cases before tackling anything more difficult, as it is obviously too difficult for you Dr., Professor, Moron. The ineptitude of your profession and the egotism you yourself show as its dimwitted ambassador speaks volumes.
You are so inept at your profession that you not only need the assistance of millions of "folding" volunteers for their collective idle cpu cycles, but also now the thousands of human participants in fold.it's "game" (the effort of which serves to help IDiots like you who are too inadequate to do the proof yourself.) Wake up, get a clue!
You're arrogant windy fellow. That's all I can say (actually I can say more, but only when the other side is open minded). I just wish that one day, when science finally adopt wholistic perspective and the evolution theory will finally be refuted as the luminiferous aether theory (and though now it seems we're far from that, it will happen) historians will document your blog as evidence to the stupidity of what will be named the secular middle ages.
Post a Comment